Forging ahead through innovation: Pfizer’s strategic progress in Q2 2025

Overview
In the second quarter of 2025, Pfizer advanced its mission to deliver breakthroughs that change patients’ lives, supported by a diversified product portfolio, innovation in R&D, and disciplined operational execution. The company navigated a dynamic healthcare landscape by strengthening its leadership in core therapeutic areas, expanding access to critical medicines, and progressing its pipeline across multiple late-stage programs.
“Pfizer had another strong quarter of focused execution and we’re pleased with our progress in advancing our R&D pipeline, driving our commercial performance and expanding our margins. We continue to strengthen our company for the future and we’re confident in our ability to create further value for patients and our shareholders”, said Dr. Albert Bourla, Chairman and CEO of Pfizer.
Q2 2025 vs. Q2 2024:
- Total revenue rose to $14.65bn (+10% YoY), driven by growth in Vyndaqel, Comirnaty, Paxlovid, Padcev, and Eliquis, partially offset by higher U.S. discounts under the IRA.
- Biopharma revenue increased to $14.31bn (+10% YoY), with growth across key therapeutic areas (cardiovascular, vaccines, oncology). Pfizer CentreOne $328m (+18% YoY) and Pfizer Ignite $20m (+38% YoY) provided incremental.
- Comirnaty was up +95% operational, with higher net revenues in the U.S. from market share gains and increased contractual deliveries in international markets.
- Paxlovid was up +71% operational, with higher net price in the U.S. and favorable adjustments to prior-period rebates, partially offset by fewer COVID infections and reduced government purchases internationally.
- Padcev was up +38% operational, with share gains in 1L la/mUC and a one-time positive impact from shifting to a wholesaler distribution model in the U.S.
- Eliquis was up +6% operational, with higher global demand, partially offset by lower net prices in the U.S. (IRA) and price erosion in certain international markets.
- Abrysvo was up +155% operational, reflecting favorable net sales adjustments and higher demand for the maternal indication in the U.S., plus launches internationally, offsetting lower older-adult vaccination after the ACIP recommendation.
- Lorbrena was up +48% operational, with greater patient share in 1L ALK+ in the U.S., China, and other markets, with U.S. pricing pressure from the IRA.
- Ibrance was down -8% operational, with lower net price in the U.S. (IRA) and generic competition, plus timing of shipments in certain international markets.
- Reported net income was $2.91bn (vs. $41m in Q2 2024), while reported EPS recorded a value of $0.51 (vs. $0.01 in Q2 2024), reflecting a favorable product mix and a lower effective tax rate versus the prior year.
- Adjusted income was $4.43bn (+30% YoY), with adjusted EPS of $0.78 (compared to $0.60 in Q2 2024), supported by commercial execution and operational efficiency.
- Reported cost of sales was $3.78bn (25.8% of revenue, +0.9 ppts YoY), with increase mainly due to the non-recurrence of a favorable royalty adjustment in Q2 2024, partially offset by lower amortization of acquired inventory.
- Reported SI&A expenses declined to $3.42bn (-8% operational), a decrease from more targeted investments and improved productivity in marketing and support functions.
- Reported R&D expenses also decreased to $2.48bn (-8% operational), driven by portfolio prioritization and lower personnel costs.
For full-year 2025, Pfizer reaffirmed its revenue guidance of $61.0bn to $64.0bn and raised its adjusted diluted EPS range by $0.10 at the midpoint to $2.90-$3.10. This updated outlook reflects strong year-to-date performance, ongoing confidence in the business, favorable foreign exchange impacts, continued progress on cost improvement initiatives, and an improved effective tax rate. Guidance also factors in a one-time $1.35bn acquired in-process R&D charge related to the licensing agreement with 3SBio, Inc., expected in the third quarter with an unfavorable impact of roughly $0.20. Additionally, the forecast accounts for the effects of current tariffs from China, Canada, and Mexico, as well as potential price changes later in the year, underscoring management’s commitment to navigating external headwinds while executing on strategic priorities.
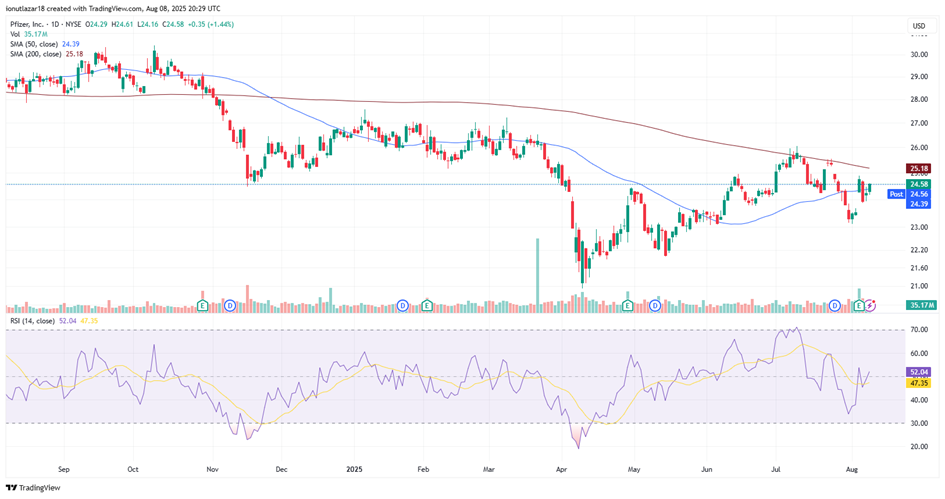
Pfizer saw a strong +5.18% jump on its Q2 earnings release day, followed by a modest -3.3% pullback in the next session, before stabilizing and closing the week with a gain of just under +5%. Year-to-date, however, the stock remains down approximately -7.6%, reflecting lingering pressure despite the recent rebound. From a technical standpoint, the price is trading slightly above its 50-day moving average ($24.39) but still below the 200-day moving average ($25.18), suggesting that a sustained bullish reversal would require a decisive breakout above the longer-term trendline. Meanwhile, the RSI at 52.04 indicates neutral momentum, not yet in overbought territory, but with room for upward movement. On the medium-to-long term, holding above the 50-day SMA and breaking the 200-day SMA could signal a shift toward a more constructive trend, while a rejection at these levels may imply further consolidation or downside risk.
Author: Ionuț-Adrian Lazar






